The type and intensity of undesirable effects are similar between SYMBICORT TURBUHALER and its components, budesonide and formoterol.No increased incidence of adverse reactions has been seen following concurrent administration of the two compounds.
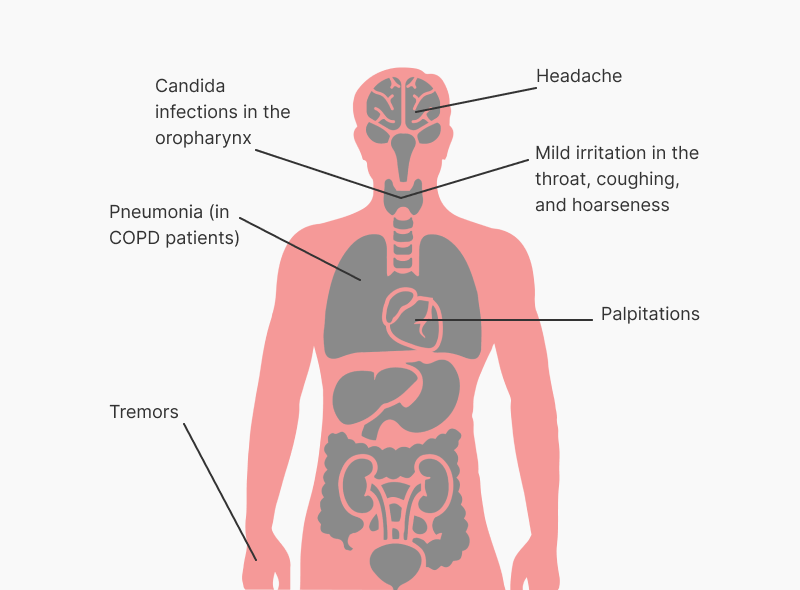
The most common drug related adverse reactions are pharmacologically predictable side-effects of beta2-agonist therapy, such as headaches, tremor, palpitations, and coughing, which tend to be mild and disappear within a few days of treatment.
The type and intensity of undesirable effects are similar between SYMBICORT TURBUHALER and its components, budesonide and formoterol.No increased incidence of adverse reactions has been seen following concurrent administration of the two compounds.
The most common drug related adverse reactions are pharmacologically predictable side-effects of beta2-agonist therapy, such as headaches, tremor, palpitations, and coughing, which tend to be mild and disappear within a few days of treatment.