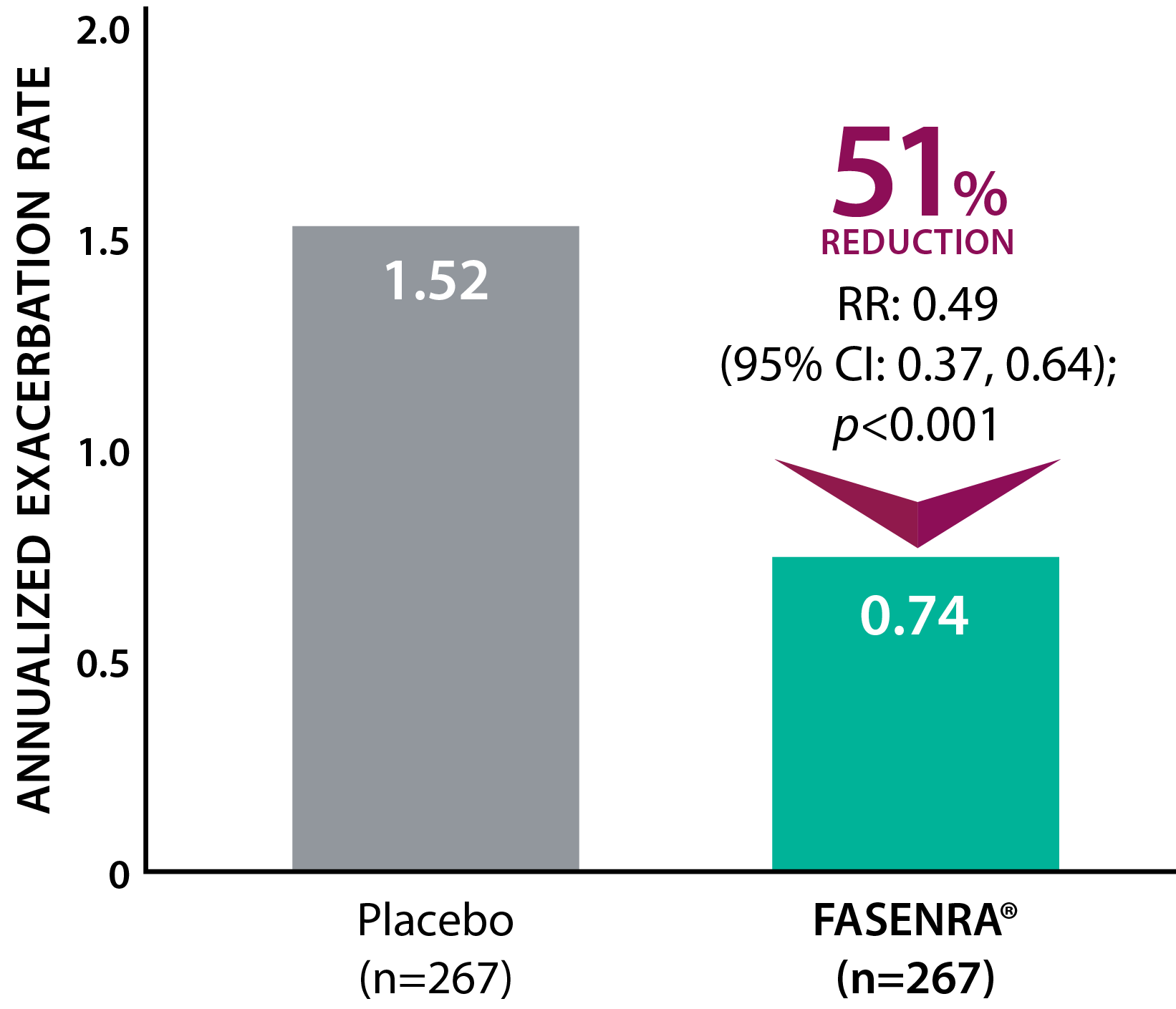
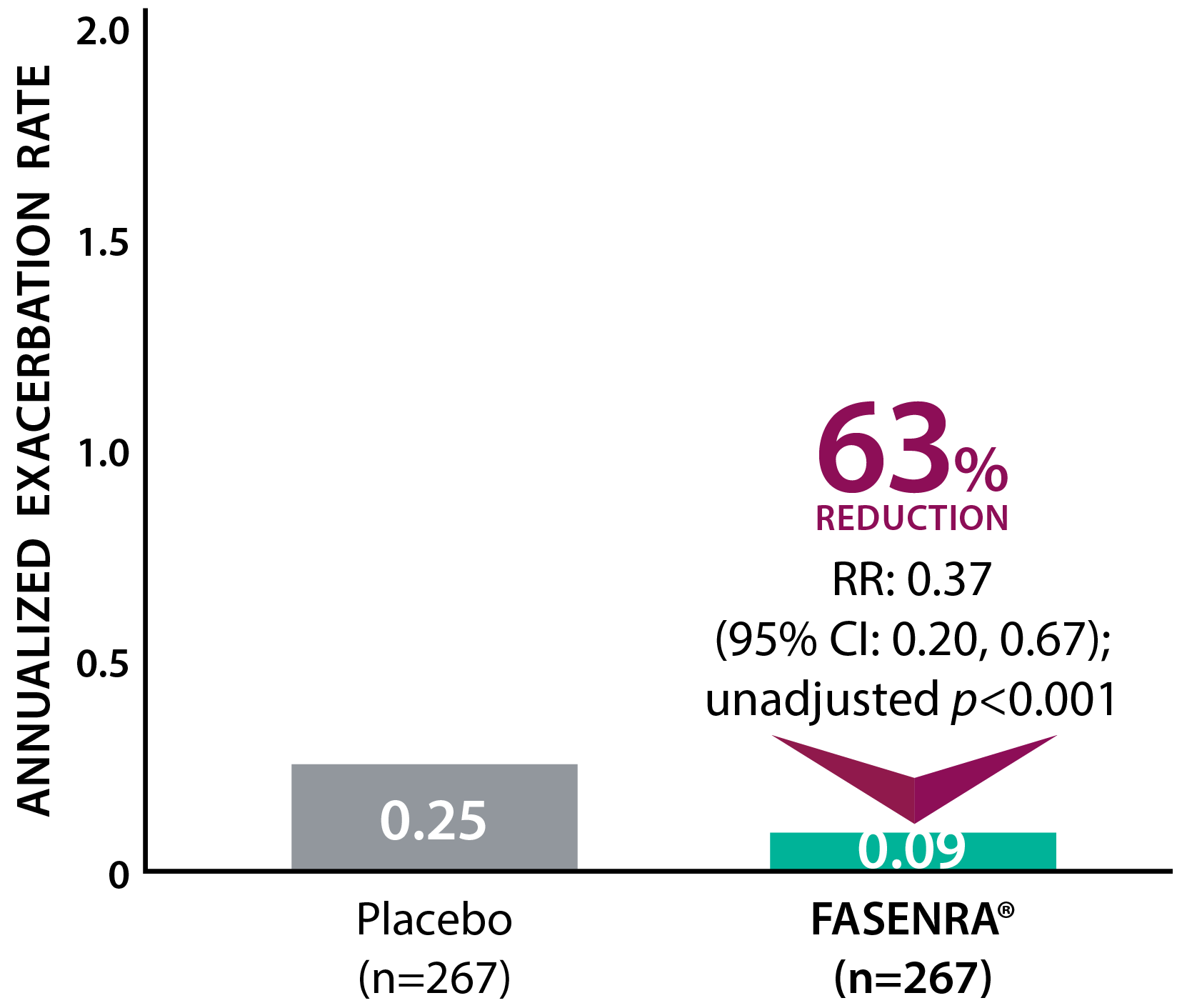
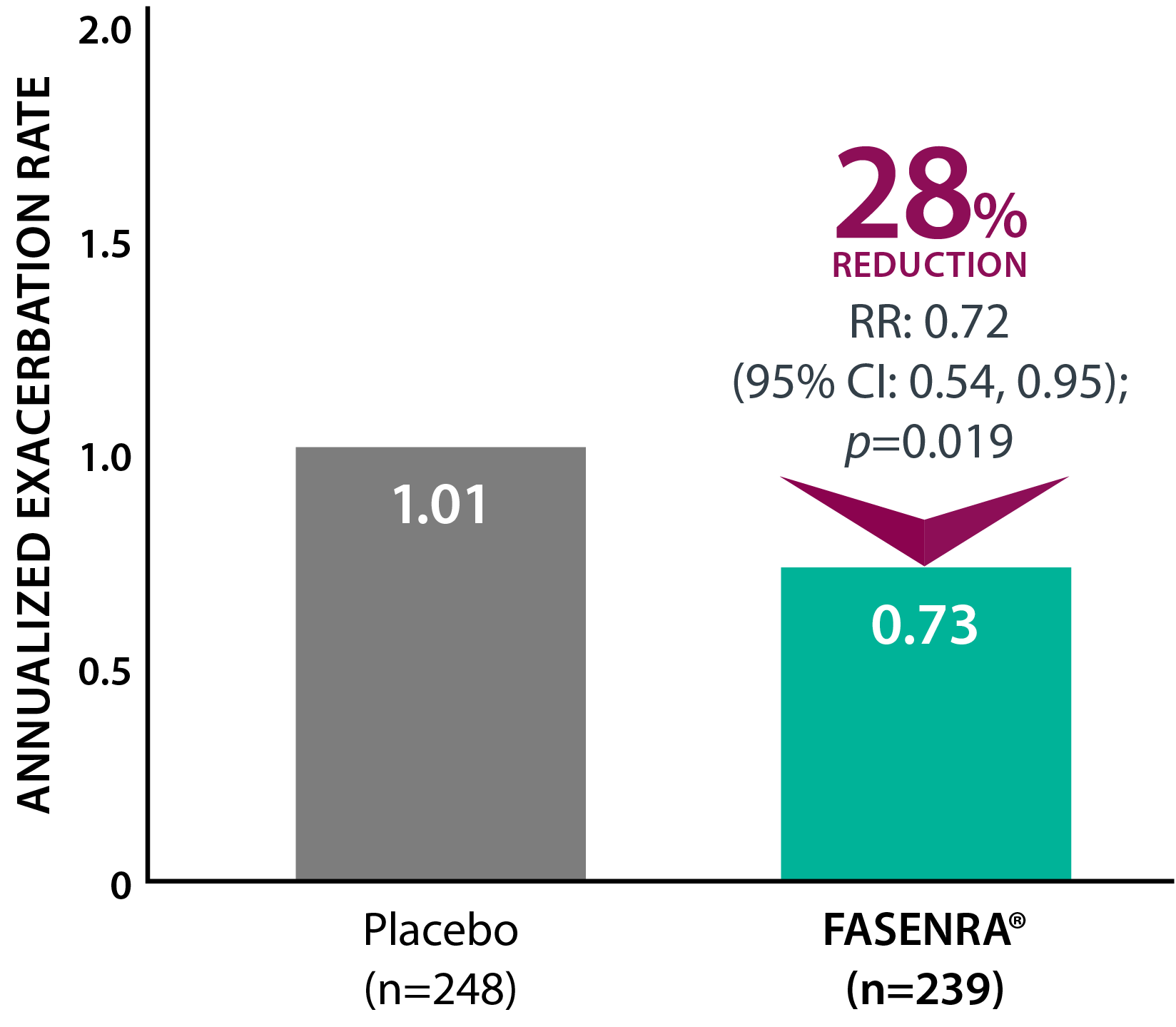
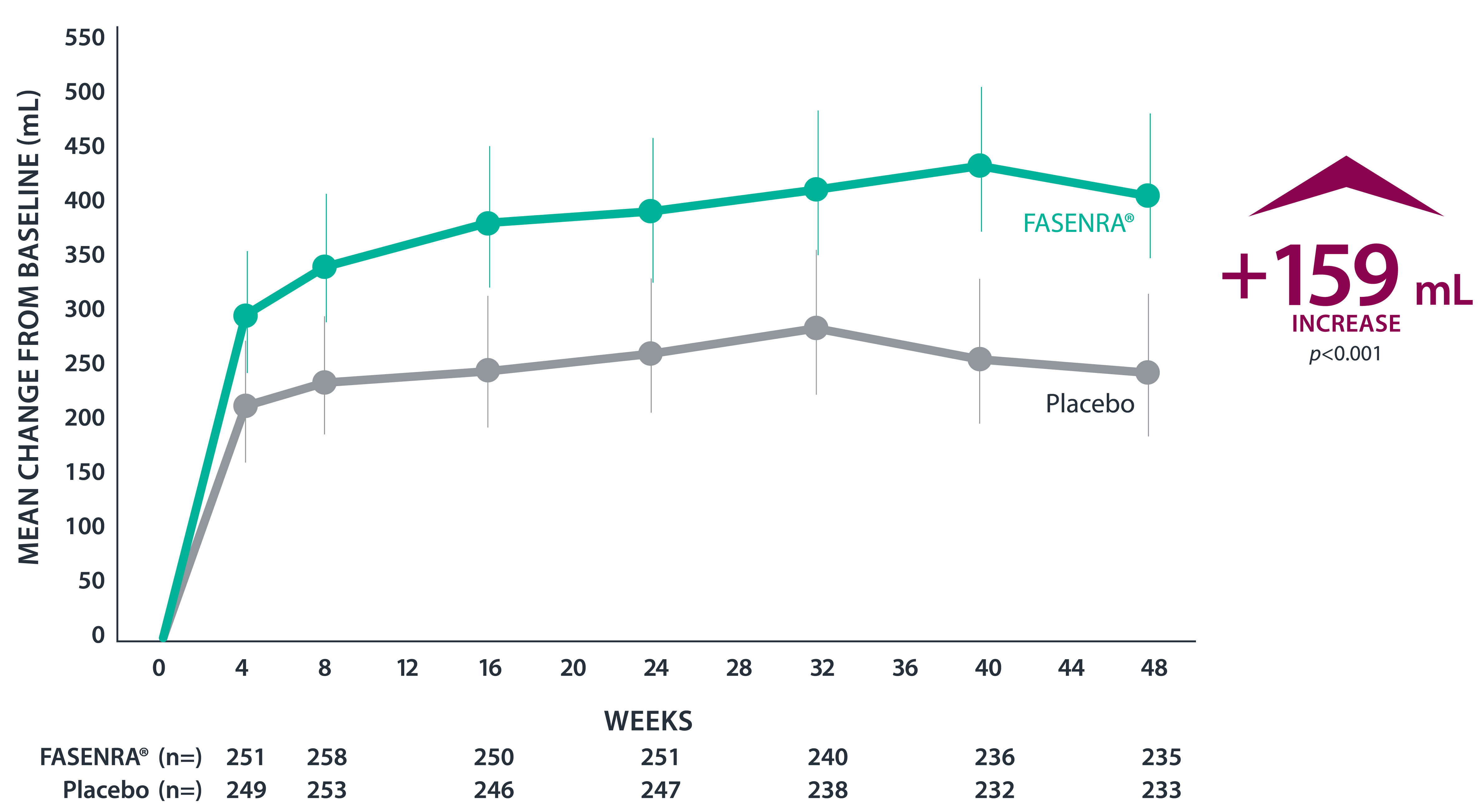
Clinically significant exacerbation = Worsening of asthma leading to use of oral/systemic corticosteroids for ≥3 days, ER visit requiring oral/systemic corticosteroids, or hospitalization. For patients on maintenance OCS, a clinically significant exacerbation requiring OCS was defined as a temporary increase in stable oral/systemic corticosteroids for ≥3 days or a single depo-injectable dose.1
* SIROCCO: 48-week, phase 3, randomized, double-blind study in patients with severe, uncontrolled asthma taking high-dose ICS + LABA. The primary efficacy (ITT) population had blood eosinophil count ≥300 cells/µL.1
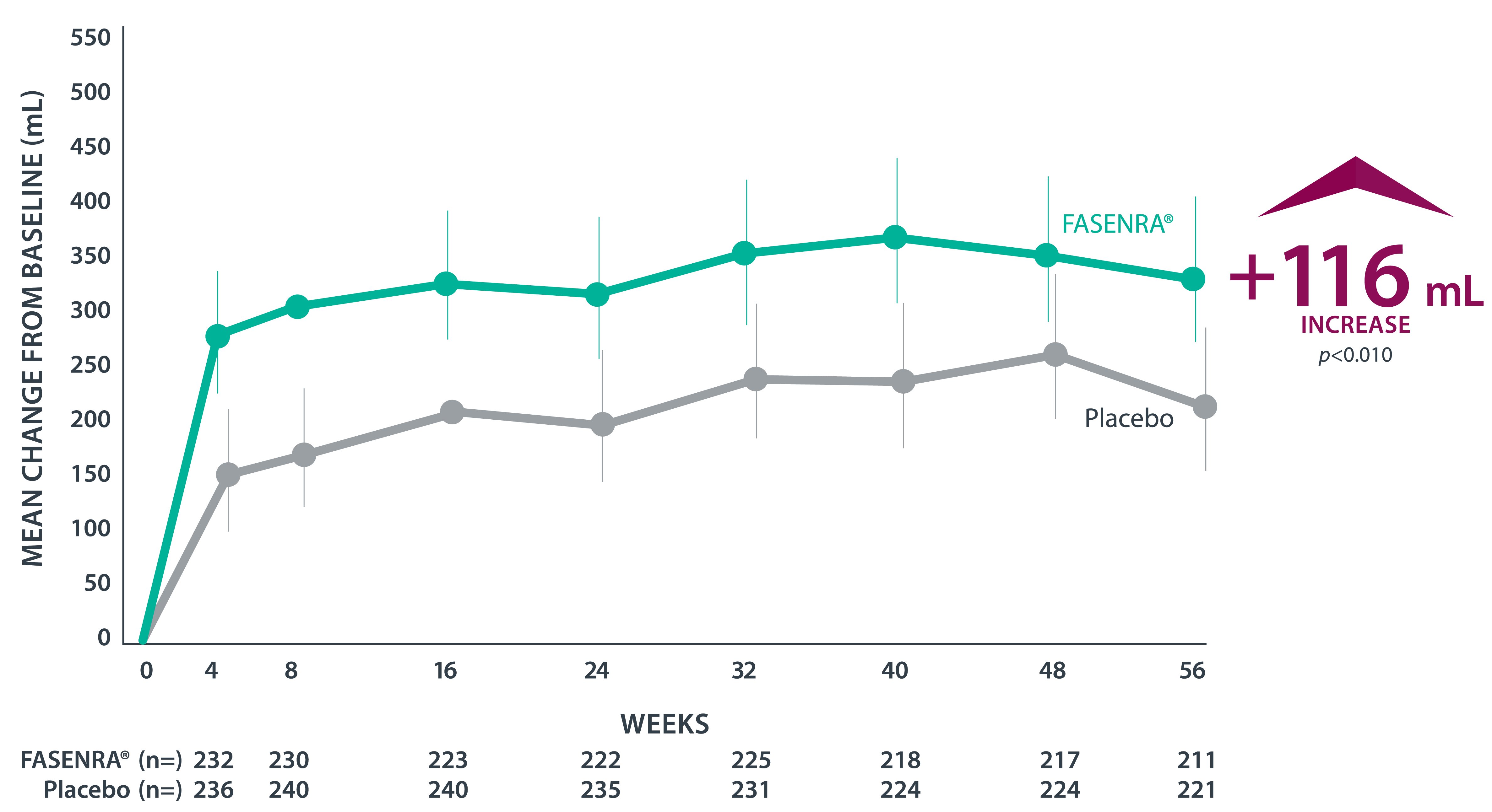
† CALIMA: 56-week, phase 3, randomized, double-blind study in patients with severe, uncontrolled asthma taking medium- or high-dose ICS + LABA. The primary efficacy (ITT) population had blood eosinophil count ≥300 cells/µL and was taking high-dose ICS.1
‡ A multinational, retrospective, observational study conducted to assess the real-world effectiveness of FASENRA®. 1002 patients with severe eosinophilic asthma were enrolled in the study and received a dose that is aligned to the Product Monograph. The study assessed annualized exacerbation rate, mOCS use, asthma symptom control, and lung function. Exacerbation was defined as worsening of asthma leading to one of the following: (1) use of systemic corticosteroid for ≥3 days or a temporary increase in a stable background dosage of OCS; (2) emergency department or urgent care visit (<24 hours) due to asthma that required systemic corticosteroid; or (3) inpatient admission to hospital (≥24 hours) due to asthma.
CI: confidence interval; ER: emergency room; ICS: inhaled corticosteroids; ITT: intent-to-treat; LABA: long-acting beta2-agonist; mOCS: maintenance oral corticosteroids; RR: rate ratio.
References:
- FASENRA® Product Monograph, AstraZeneca Canada Inc.
- Bleecker ER, FitzGerald JM, Chanez P, et al. Efficacy and safety of benralizumab for patients with severe asthma uncontrolled with high-dosage inhaled corticosteroids and long-acting
β2-agonists (SIROCCO): a randomised, multicentre, placebo-controlled phase 3 trial. Lancet. 2016;388(10056):2115–2127 (incl. supplement).
- Jackson DJ, Pelaia G, Emmanuel B, et al. Benralizumab in severe eosinophilic asthma by previous biologic use and key clinical subgroups: real-world XALOC-1 programme. Eur Respir J. 2024;2301521.